BY EBELE ORAKPO with Agency Reports
MALIGNANT neoplasia, popularly called cancer, is said to kill about 10,000 Nigerians yearly. Whether this is true or not is not the issue. The fact still remains that many die from various types of cancer annually but the good news is that succour is coming the way of sufferers through the work of some biophysicists at Rice University, USA-based Center for Theoretical Biological Physics (CTBP).
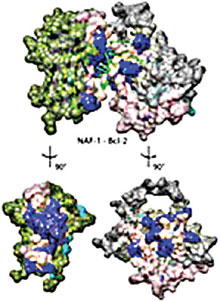
The researchers have uncovered a previously unknown binding interface between two key proteins involved in a cell’s decision to survive or die. Using computer analysis of experimental data, CTBP researchers discovered that the way B-cell lymphoma 2 (Bcl-2) and Nuclear Assembly Factor 1 Ribonucleoprotein (NAF-1) interact, is a major determinant in the cell processes of autophagy and apoptosis (life and death).
As experts have discovered, cancer involves unregulated cell growth which means that cells divide and grow uncontrollably. They no longer obey the laws as it were. They just keep growing and forming malignant tumors , which most times, invade other parts of the body and eventually lead to death.
According to the report published in the Proceedings of the National Academy of Sciences, Bcl-2 regulates cell death (apoptosis ), by either inducing (pro-apoptotic) it or inhibiting it (anti-apoptotic). Any damage to the Bcl-2 gene has been identified as a cause of a number of cancers . NAF-1 on the other hand, a member of the NEET family that binds toxic clusters of iron and sulfur, was shown to be a key player in regulating autophagy.
Cellular components
Autophagy helps maintain a balance between manufacture of cellular components and break down of cellular constituents.
“NAF-1 binds to two regions of Bcl-2, and Bcl-2 binds to the NAF-1 groove between the beta cap and iron-sulfur cluster binding domains, which is the heart of the strongest binding domain, meaning that Bcl-2 interaction affects NAF-1 activity,” the report said.
“The ability to block a specific binding site or to enhance a desired interaction is critical to drug design,” said José Onuchic, professor of physics and astronomy at Rice University. The scientists believe that this discovery could lead to new medications.
“The binding interface can be interrogated for medicinal purposes using medicines that bind either protein, as certain binding sites of the proteins can send a cell toward apoptosis or survival. Knowing which binding site causes what fate is critical for drug design,” he said, adding: “In our early work, we have shown the link between NEET proteins and cancer.
Now we can understand the molecular details of how these interactions are governed. It has been shown that NAF-1 is up-regulated in cancer cells, which leads us to believe that cancer may hijack control over the expression of this protein. This affects the cell’s system of checks and balances. Understanding NAF-1 gives us a better idea of how to approach treatment,” he said.
The researchers found that NAF-1 binds to two specific regions of the protein Bcl-2 and that Bcl-2 binds to the NAF-1 groove formed between the beta cap and iron-sulfur cluster binding domains; the strongest coupling is at the cluster binding domain and some contacts of interest are at the top of the beta-cap domain.
Iron-sulfur cluster
Since the iron-sulfur cluster is the functional entity involved in NAF-1 activity, these findings clearly indicate that Bcl-2 interaction with NAF-1 affects its activity, Onuchic said.
The researchers performed functional studies of cluster transfer and other full-length protein interactions with a spectrometer sensitive to hydrogen/deuterium exchange using a combination of experimental and theoretical methods. They combined their results with a computer-based process created at CTBP called direct coupling analysis (DCA).
Said Patricia Jennings, co-lead author and professor of chemistry and biochemistry at CTBP affiliate based at the University of California, San Diego: “Each of the three techniques not only confirmed the results of the other methods but also provided unique insights in their own right,” noting that the combined techniques are applicable to biomolecular interactions in general.
“DCA helps us efficiently filter through massive amounts of data and does not require high-resolution structural studies, although those are desirable,” she said. “Peptide array is powerful for localizing fragments that bind with high affinity, and hydrogen/deuterium exchange studies allow us to monitor parts of the intact protein that are not seen in structural studies and are not amenable to DCA analysis. “Together, the techniques provide an exquisite synergy,” she said.
Other members of the team are Rachel Nechushtai, professor at the Hebrew University of Jerusalem; Assaf Friedler, a professor at the Hebrew University Institute of Chemistry; and Ron Mittler, a professor of biological sciences at the University of North Texas, Denton. The team had in a previous research identified NAF-1 as one of two prime suspects in the proliferation of breast cancer.
Disclaimer
Comments expressed here do not reflect the opinions of Vanguard newspapers or any employee thereof.